For three decades, hantavirus lived in the category Wall Street usually ignores: deadly, terrifying, but too rare to monetize. Then six cruise ship passengers got infected. And within days, Moderna shares surged nearly 19%.
Moderna Stock Reaction
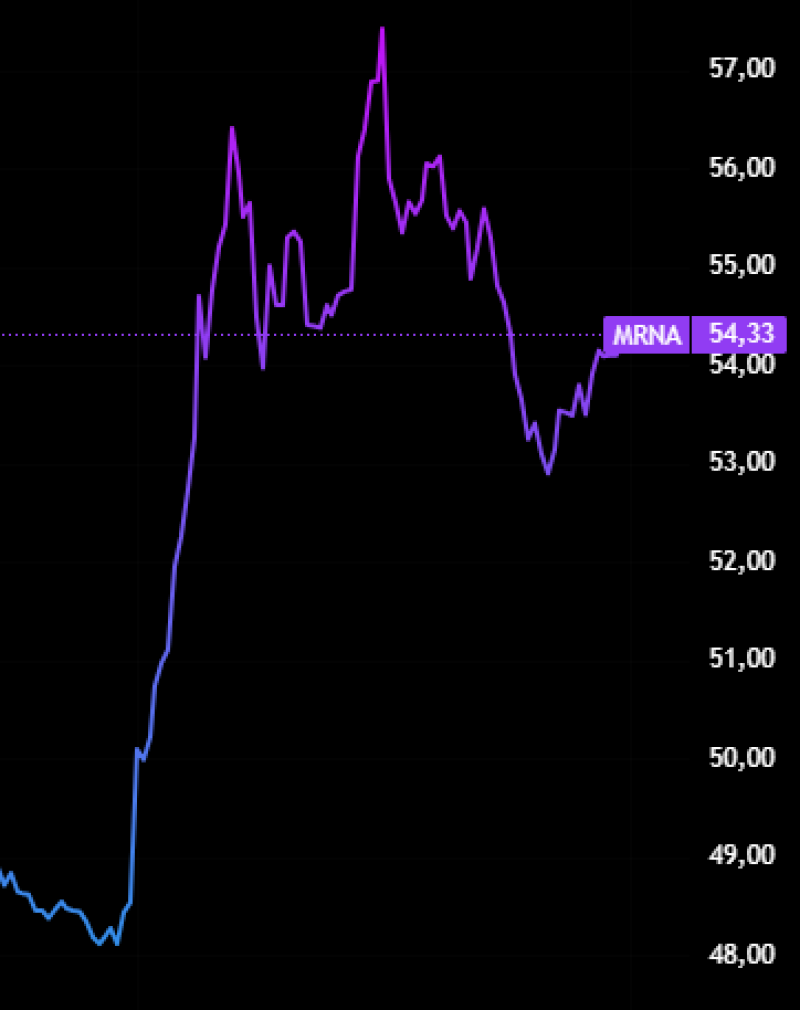
Moderna shares jumped sharply after markets began pricing in the long-term value of its mRNA biodefense platform rather than the commercial potential of a single hantavirus vaccine.
At first glance, the reaction looked irrational. Hantavirus pulmonary syndrome has caused only around 890 confirmed US cases in the US between 1993 and 2023. That is fewer than 30 infections annually. Even with a mortality rate approaching 38%, the numbers are far too small to justify a traditional vaccine business.
So why did investors suddenly care?
Because the market is no longer valuing Moderna as a vaccine company. It is starting to value it as a biological operating system.
The Old Vaccine Model Never Worked for Rare Diseases
Under the traditional pharmaceutical model, developing a vaccine is brutally expensive. Companies often spend $1–2 billion and wait close to a decade before approval. That equation only works when the final market is massive.
Hantavirus never fit that math.
Neither did Ebola for many years. Nor Marburg. Nor Nipah.
These pathogens were considered strategically important but commercially unattractive. Shareholders do not reward companies for spending billions to solve diseases that may infect only dozens of people annually. That is why hantavirus remained largely untouched for decades despite its frightening lethality.
The disease attacks the lungs rapidly, often progressing into severe respiratory failure. Nearly four out of every ten infected patients die. Yet the total addressable market remained too small for conventional biotech economics.
Moderna Changed the Cost Structure
The reason investors reacted this week has little to do with hantavirus itself. It has everything to do with what mRNA platforms change about vaccine development.
Moderna confirmed that it is advancing hantavirus research through partnerships with the US Army Medical Research Institute of Infectious Diseases (USAMRIID) and Korea University’s Vaccine Innovation Center. The programs remain early-stage and are not yet funded for human trials.
But investors were not buying near-term revenue projections from a hantavirus shot. They were buying the idea that Moderna can pursue dozens of niche pathogen programs simultaneously because the underlying infrastructure already exists.
The expensive part of vaccine development used to be building the machinery from scratch every time:
- new manufacturing systems
- new delivery approaches
- new regulatory pathways
- new production validation
mRNA compresses much of that process into a reusable platform. Once the “factory” exists, adding another pathogen target becomes dramatically cheaper. The marginal cost of developing additional vaccines drops sharply compared to traditional approaches.
That changes the investment equation entirely.
Instead of asking:
Can this single vaccine become a blockbuster product?
Markets are starting to ask:
How many pathogens can this platform respond to at once?
The Real Customer May Not Be Consumers
Another reason the hantavirus story matters is that rare-pathogen vaccines often do not rely on ordinary commercial demand. Governments do not buy biodefense capacity the same way consumers buy flu shots.
Organizations like BARDA, CEPI, and military biodefense agencies pay for preparedness:
- manufacturing readiness
- rapid response capability
- strategic stockpiles
- emergency deployment infrastructure
That means even diseases with tiny infection counts can still generate meaningful contracts. In this model, rare outbreaks function less like traditional healthcare markets and more like national security events.
The MV Hondius cruise ship outbreak appears to have acted as the catalyst that forced markets to reprice this reality. Moderna’s research began before it, but the outbreak reminded investors how quickly obscure pathogens can become headline risks.
Every emerging infectious cluster now becomes a potential validation event for platform-based biotech companies.
Moderna’s Core Business Is Also Stabilizing
The timing also mattered because Moderna’s financial picture has recently improved.
The company reported Q1 2026 revenue of $389 million, up 260% year over year. Earnings per share significantly exceeded expectations, while its flu vaccine candidate delivered positive Phase 3 efficacy data against standard flu shots. That matters because investors had spent much of the post-pandemic period questioning whether Moderna could survive beyond COVID vaccine demand.
Now the narrative is shifting.
Instead of being viewed as a one-product pandemic winner, Moderna is increasingly being priced as a scalable biotechnology platform with applications across infectious disease, cancer therapeutics, and biodefense.
Every additional program potentially lowers the average cost of future development because the same manufacturing, research, and regulatory infrastructure can be reused repeatedly. In software terms, Moderna is beginning to resemble a platform company more than a traditional pharmaceutical firm.
What Investors Are Actually Buying
The hantavirus program itself may never become a major standalone revenue source. That is not the point. The market reaction reflects something larger: the belief that mRNA technology can turn previously “unprofitable” diseases into economically viable targets.
The platform has theoretically unlimited applications:
- emerging viruses
- military biodefense
- pandemic preparedness
- personalized cancer vaccines
- rapid-response outbreak systems
The more outbreaks appear globally, the more valuable rapid-response biological infrastructure becomes. And that may explain why six infections on a remote cruise ship were enough to move billions of dollars in market value.
Investors were not buying a hantavirus vaccine. They were buying optionality on a future where every outbreak becomes monetizable infrastructure demand.
 Peter Smith
Peter Smith
 Peter Smith
Peter Smith


